CLEEN Cleaning Validation Module
CLEEN is built by experts who’ve lived the challenges of contamination control, compliance and pharma manufacturing – embedding real-world experience into every feature.

Embracing Pharma 4.0
Why operational digitization must be the top priority for pharma and biopharma manufacturers.
This free eBook outlines the fundamental shifts associated with embracing Pharma 4.0 — and how they can empower organizations to improve efficiency, agility, and maintain product quality while driving business profitability.
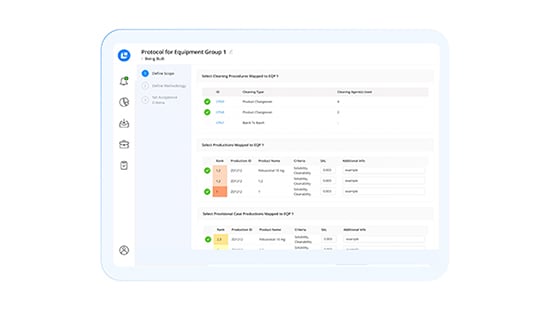
Modernize Cleaning Validation With Intelligent Automation
Add a Descriptive and SEO Subtitle Here
Ensure Compliant Validations
Receive in-platform guidance as you build cleaning validations based on continuously updated regulations. Automatically calculate carryover limits and worst-case product rankings, and leverage automated compliance interlocks for CHT/DHT.
Accelerate Re-Validation
Leverage historical data to calculate carryover limits and create protocols in minutes by integrating data from multiple sources, speeding up new product introductions. Receive dynamic alerts for necessary re-validations complete with auto-generated documents for review.
Simplify Audit-Readiness
Automatically generate validation reports and store all necessary information in the built-in Audit Portal for immediate access that enables continuous audit-readiness with minimal administrative burden.
Key Features
Ensure Accurate HBEL-based Residue Limit Calculations
Health-Based Exposure Limits (HBELs) are scientifically derived thresholds that define the maximum acceptable carryover of pharmaceutical residues on shared manufacturing equipment.
- Calculate safe residue limits automatically using HBEL data to ensure worst-case MACO (maximum allowable carryover) compliance.
- CLEEN supports risk-based cleaning validation in pharma aligned with FDA and ICH expectation.

New Drug Introduction Risk Assessment
Evaluate the implications of integrating new drugs into current production lines. Instantly determine carryover thresholds, risk levels, and worst-case scenarios. CLEEN’s risk matrix supports cleaning validation, ensures regulatory compliance and enhances operational efficiency.
- Instant impact analysis to assess cross-contamination risks in shared facilities.
- Analytical method validation, especially around Limit of Detection (LOD).
- Forecasting future risks and updating cleaning validation parameters.

Auto-Generate Standardized, Compliant Cleaning Validation Protocols
- Auto-generates protocols using built-in risk assessments, HBEL data and historical residue profiles.
- Supports carryover limit calculations for actives, detergents, microbials and nitrosamines.
- Enables one-click protocol creation by aggregating risk assessments, analytical methods and toxicity data.
- Provides real-time alerts when new validations are required, ensuring compliance continuity.

Integrate Residue Data from LIMS to Generate Reports
- Imports residue results from LIMS directly into the validation platform.
- Generates comprehensive reports using pre-configured templates.
- Supports automated protocol generation, residue limit calculations and real-time validation tracking.

Track Real-Time Validation Status for all Equipment
- Tracks real-time validation status across all equipment.
- Visually distinguishes between completed, ongoing and pending validations.
- Integrates with scheduling tools to manage multiple studies simultaneously.
- Supports customizable reports for FDA.

Leverage Audit Portal During Inspections
- Immediate access to validation reports, SOPs, protocols and supporting documents.
- Digitally-signed, Part 11-compliant logs for traceability and regulatory alignment.
- Maintain a complete audit trail of changes, validations and operator actions.
- Allows external auditors to securely access relevant data without exposing unrelated systems.
- Regulatory Inspections: Pull up product-specific reports for a 360° view of cleaning performance.

Frequently Asked Questions
Compliant and Scalable Cloud Infrastructure
CLEEN automates MACO calculations, residue limit assessments, and standardized reporting to align with FDA, EMA, and ICH cleaning validation standards. Its audit-ready documentation ensures you’re always inspection-prepared.
Yes. CLEEN is built to integrate seamlessly with your current Quality Management Systems, Laboratory Information Management Systems, and ERPs – eliminating data silos and ensuring a connected validation ecosystem.
CLEEN offers end-to-end automation, real-time tracking, and configurable workflows designed specifically for pharmaceutical industry. It’s built with audit-readiness and regulatory alignment at its core – no generic solutions here.
Absolutely. CLEEN is fully compliant with 21 CFR Part 11, offering secure audit trails, electronic signatures, role-based access control, and data encryption to meet all global data integrity requirements.
CLEEN allows you to assess risks based on product toxicity, solubility, batch size, and more, helping to prevent cross contamination. It calculates worst-case scenarios automatically and supports ICH and APIC cleaning validation frameworks.
Customers typically reduce manual validation effort by up to 60%, speed up audit prep by 80%, and reduce compliance risks significantly. CLEEN also saves costs by streamlining documentation and reducing rework.
Implementation time depends on your facility size and system integrations. Most clients are fully operational within 4-8 weeks, with support from our dedicated onboarding and validation teams.
Yes. CLEEN is a secure, cloud-based platform accessible from anywhere, enabling real-time collaboration across global teams while maintaining regulatory compliance.
We do. CLEEN includes hands-on onboarding, live training sessions, detailed documentation, and 24/7 customer support to ensure your team gets up to speed quickly and confidently.
Absolutely. You can request a personalized demo tailored to your facility’s needs. We’ll show how CLEEN fits into your existing workflows and regulatory strategy.


